PRODUCT MONOGRAPH
PLAN B
®
Levonorgestrel Tablet
1.5 mg
Manufacturer’s Standard
EMERGENCY CONTRACEPTION
Foundation Consumer Healthcare, LLC
1190 Omega Drive
Pittsburgh, PA 15205. USA
Paladin Labs Inc. Date of Preparation:
100 Blvd. Alexis Nihon, Suite 600
Saint-Laurent, Quebec April 26, 2018
H4M 2P2 Version 12.0
Submission Control No: 214131

Page 2 of 36
Table of Contents
PART I: HEALTH CARE PROFESSIONAL INFORMATION ............................................ 3
SUMMARY PRODUCT INFORMATION ........................................................................ 3
INDICATIONS AND CLINICAL USE ............................................................................. 3
CONTRAINDICATIONS ................................................................................................... 4
WARNINGS AND PRECAUTIONS ................................................................................. 4
ADVERSE REACTIONS ................................................................................................... 7
DRUG INTERACTIONS.................................................................................................. 11
DOSAGE AND ADMINISTRATION ............................................................................. 13
OVERDOSAGE ................................................................................................................ 13
ACTION AND CLINICAL PHARMACOLOGY ............................................................ 14
STORAGE AND STABILITY ......................................................................................... 16
DOSAGE FORMS, COMPOSITION AND PACKAGING ............................................. 16
PART II: SCIENTIFIC INFORMATION ................................................................................ 17
PHARMACEUTICAL INFORMATION ......................................................................... 17
CLINICAL TRIALS ......................................................................................................... 18
DETAILED PHARMACOLOGY .................................................................................... 24
TOXICOLOGY ................................................................................................................. 25
REFERENCES .................................................................................................................. 29
PART III: CONSUMER INFORMATION ............................................................................. 32
Plan B - Product Monograph Page 3 of 36
PLAN B
®
Levonorgestrel 1.5 mg Tablet
PART I: HEALTH CARE PROFESSIONAL INFORMATION
SUMMARY PRODUCT INFORMATION
Route of
Administration
Dosage Form /
Strength
Clinically Relevant Non-Medicinal
Ingredients
Oral
Tablet 1.5 mg
Lactose Monohydrate
For a complete listing see Dosage Forms,
Composition and Packaging section
INDICATIONS AND CLINICAL USE
Plan B
®
(levonorgestrel 1.5 mg tablets) is an emergency contraceptive that can prevent
pregnancy and is intended to be used within 72 hours (three days) following known or suspected
contraceptive failure or unprotected intercourse such as:
when no contraceptive has been used
when a contraceptive method may have failed, including:
o condom rupture, slippage, or misuse
o diaphragm or cap dislodgment, breakage or early removal
o failed coitus interruptus
o miscalculation of periodic abstinence method
o expelled intrauterine device
o missed oral contraceptive
o a delay in starting a new packet of oral contraceptives
o a delay in getting a scheduled contraceptive injection
in cases of sexual assault
Treatment should not be delayed as efficacy may decline with an increased interval between
intercourse and the start of the treatment. Efficacy is greatest when treatment is taken within 24
hours of unprotected intercourse, decreasing somewhat during each subsequent 24-hour period.
The pregnancy rate of Plan B
®
(levonorgestrel 1.5 mg tablet) is calculated for a single use. If Plan
B
®
is used on more than one occasion, the cumulative pregnancy rate will be higher. Plan B
®
is
not recommended for routine use as a contraceptive.
Plan B
®
will not prevent pregnancy from future acts of unprotected intercourse. Following use of
this product, the woman should either abstain or use an alternative contraceptive method until her
next menstrual cycle.

Plan B - Product Monograph Page 4 of 36
Note to Pharmacist: If you determine that a woman is a repeat user of Emergency Contraception
(defined as use more than once a month on a regular basis) or that Plan B
®
has been used within
the past cycle, you should consider discussing other, more effective contraceptive methods with
the woman, as well as encouraging her to see her physician or other health professional for
contraceptive counselling services and advise on other methods of contraception and prevention
of sexually transmitted infections. Plan B
®
should still be dispensed, if indicated.
Geriatrics: Plan B
®
has not been studied in this population.
Pediatrics: Plan B
®
has not been studied in this population.
CONTRAINDICATIONS
Patients who are hypersensitive to the active substance levonorgestrel or to any ingredient in
the formulation. For a complete listing, see the Dosage Forms, Composition and Packaging
section of the product monograph.
Women with known or suspected pregnancy. The method is not to be used by a woman who
is pregnant due to a previous act of intercourse, especially if there is recent abnormal
bleeding, a pregnancy test should be performed before taking Plan B.
Patients with undiagnosed abnormal vaginal bleeding, a pregnancy test should be performed
before taking Plan B.
Progestin-only oral contraceptives are used as a routine method of birth control over longer
periods of time, and are contraindicated in some conditions (acute liver disease or history of or
actual benign or malignant liver tumours, known or suspected carcinoma of the breast, and
undiagnosed abnormal vaginal bleeding). It is not known whether these same conditions apply
to the Plan B
®
regimen consisting of the emergency use of one progestin pill, but these risks
should be considered if Plan B
®
needs to be administered several times.
WARNINGS AND PRECAUTIONS
General
Plan B
®
is not an abortifacient and should not be taken by pregnant women, as it will not
be effective.
Patients should be advised that Plan B
®
provides no protection against HIV infection
(AIDS) and other sexually transmitted diseases.
The use of cyclic combination oral contraceptives containing estrogen and progestin is
associated with increased risks of several serious conditions, including thromboembolic and
cardiovascular disorders (e.g. thrombophlebitis, pulmonary embolism, cerebrovascular disorders,
myocardial ischemia, mesenteric thrombosis, retinal thrombosis), hepatic neoplasia and

Plan B - Product Monograph Page 5 of 36
gallbladder disease. These conditions have not been associated with the routine use of progestin-
only oral contraceptives, but whether short-term (single dose) use of high-dose progestin-only
contraceptives would accentuate the risk is unknown. Plan B
®
does not contain estrogen.
Controlled clinical trials using Plan B
®
and post-market experience with levonorgestrel for post
coital and emergency contraception have not so far identified any serious adverse events.
Plan B
®
is not recommended for routine use as a contraceptive. The pregnancy rate of Plan
B
®
(levonorgestrel 1.5 mg tablet) is calculated for a single use. If Plan B
®
is used on more than
one occasion, the cumulative pregnancy rate will be higher.
Migraine and Headache: During the use of Plan B
®
, the onset or exacerbation of migraine or
the development of a new pattern that is recurrent, persistent or severe requires evaluation of the
cause and may require re-evaluation of a future use of emergency contraceptive pills.
Concomitant use of Plan B and drugs containing ulipristal acetate is not recommended (see Drug-
Drug Interactions section).
Carcinogenesis and Mutagenesis
See Toxicology section in Part II: Scientific Information.
Cardiovascular
Hypertension: Patients with essential hypertension whose blood pressure is well controlled may
be given combined oral contraceptives containing estrogen and progestin, but only under close
supervision. Progestin-only oral contraceptives are not contraindicated for such patients.
Endocrine and Metabolism
Diabetes: The effects of Plan B
®
on carbohydrate metabolism are unknown. Some users of
progestin-only oral contraceptives experience slight deterioration in glucose tolerance, with
increases in plasma insulin; however, women with diabetes mellitus who use progestin-only oral
contraceptives do not generally experience changes in their insulin requirements. Nevertheless,
diabetic women should be monitored while taking Plan B
®
.
Hepatic
Hepatic function: Following a single oral dose of 0.75 mg, levonorgestrel does not appear to be
significantly metabolized by the liver. The risks of Plan B
®
to women with a history of liver
disease are unknown. Women with a history of liver disease should be given Plan B
®
under
medical supervision especially if the method needs to be administered more than once.

Plan B - Product Monograph Page 6 of 36
Sexual Function/Reproduction
Effects on Menses: Menstrual bleeding patterns are often irregular among women using
progestin-only oral contraceptives and in clinical studies of levonorgestrel for postcoital and
emergency contraceptive use. Some women may experience spotting a few days after taking
Plan B
®
. At the time of expected menses, approximately 77% of women using Plan B
®
had
vaginal bleeding similar to their normal menses, 11-12% bled more than usual, and 11% bled
less than usual. The majority of women (78%) had their next menstrual period at the expected
time or within 5 days, while only 4.5% had a delay of more than 7 days beyond the anticipated
onset of menses. If there is a delay in the onset of menses beyond 1 week, the possibility of
pregnancy should be considered.
Ectopic Pregnancy: Ectopic pregnancies account for approximately 2% of reported
pregnancies (19.7 per 1000 reported pregnancies). Up to 10% of pregnancies reported in
clinical studies of routine use of progestin-only oral contraceptives are ectopic. However,
there appears to be no increase in the rate of ectopic pregnancy after use of levonorgestrel for
emergency contraception. A history of ectopic pregnancy need not be considered a
contraindication to use of this emergency contraceptive method. However, physicians should be
alert to the possibility of an ectopic pregnancy in women who become pregnant or complain of
lower abdominal pain after taking Plan B
®
.
Suspected Pregnancy: A pregnancy test is warranted if pregnancy is suspected. Women
should be counselled to abstain from sexual intercourse or use an alternative contraceptive
method until the onset of their next normal menstrual period. If a normal menstrual period is
delayed beyond 1 week, the patient’s pregnancy status should be confirmed with a pregnancy
test and follow-up with a health professional. Counselling on routine contraception for future
use should be provided as appropriate.
Special Populations
Pregnant Women: Plan B
®
is not an abortifacient and should not be taken by pregnant women,
as it will not be effective. Studies involving women who have taken combined oral
contraceptives containing levonorgestrel inadvertently during early pregnancy do not suggest
that these drugs have an adverse effect on the fetus and there is no evidence that Plan B
®
(levonorgestrel 1.5 mg tablet) taken as an emergency contraceptive would have an adverse effect
on an established pregnancy. However, there are insufficient data to rule out the possibility of
adverse effects on the fetus if Plan B
®
is used after a woman is already pregnant or in cases of
method failure.
Nursing Women: Administration of combined oral contraceptives and progestin-only
contraceptives to breastfeeding women has been reviewed in the literature
13
. Seven studies were
reviewed that analyzed the transmission of progestins in breast milk. Data were obtained as
early as one week post-partum up to approximately six months post-partum. Very small
amounts of progestin have been measured in the milk of breastfeeding mothers who are taking
progestin-only contraceptives. Levonorgestrel is transferred from maternal breast milk to

Plan B - Product Monograph Page 7 of 36
infants, with infant plasma levels approximately 40% of those in breast milk and approximately
1% to 6% of maternal plasma
13, 14
. No adverse effects due to progestin-only oral contraceptives
have been found on breastfeeding performance, either in the quality or quantity of the milk, or on
the health, growth, or development of the infant.
Pediatrics: No data is available.
Geriatrics: No data is available.
Body weight: Limited and inconclusive data suggest that there may be reduced efficacy of
levonorgestrel with increasing body weight or body mass index (BMI). In all women, emergency
contraception should be taken as soon as possible after unprotected intercourse, regardless of the
women’s body weight or BMI.
ADVERSE REACTIONS
Adverse Drug Reaction Overview
There are in general no serious adverse drug reactions to report following the use of Plan B
®
,
either in clinical trials or post-market surveillance. Most commonly observed adverse drug
reactions are presented in the following sections.
Clinical Trial Adverse Drug Reactions
Because clinical trials are conducted under very specific conditions the adverse reaction rates
observed in the clinical trials may not reflect the rates observed in practice and should not be
compared to the rates in the clinical trials of another drug. Adverse drug reaction information
from clinical trials is useful for identifying drug-related adverse events and for approximating
rates.
Plan B
®
divided dose regimen
The most common adverse events reported in the Pivotal Study of Plan B
®
(Study 92908)
(levonorgestrel 0.75 mg tablets) when administered as two doses of 0.75 mg at a 12-hour interval
included:
Nausea: 23%
Abdominal pain: 18%
Fatigue: 17%
Headache: 17%
Vomiting: 6%
Intermenstrual bleeding and altered menstrual cycles: Some women may experience
spotting a few days after taking Plan B
®
. The majority of women (58%) will have their
next menstrual period at about the expected time or a few days early or late; if there is a
Plan B - Product Monograph Page 8 of 36
delay in the onset of menses of more than one week, the possibility of pregnancy should
be excluded. Women who take Plan B
®
frequently are likely to experience disruptions of
the menstrual cycle.
Other: breast tenderness, dizziness and diarrhea have been reported in women using Plan
B
®
and may be drug related.
In this comparative clinical study involving 1,955 evaluable women, the incidence of nausea and
vomiting, were significantly (P<0.01) less for women using Plan B
®
than for women receiving
the Yuzpe regimen. Adverse events reported in the other controlled clinical trial of the Plan B
®
regimen by Ho and Kwan
10
were consistent with those in the Pivotal Study (Study 92908)
(Table 1).
In the combined controlled clinical trials, the proportion of women receiving levonorgestrel who
reported nausea was less than half of the proportion in the Yuzpe group (Table 2). The
proportion that reported vomiting in the levonorgestrel group was only one-fourth that in the
Yuzpe group.
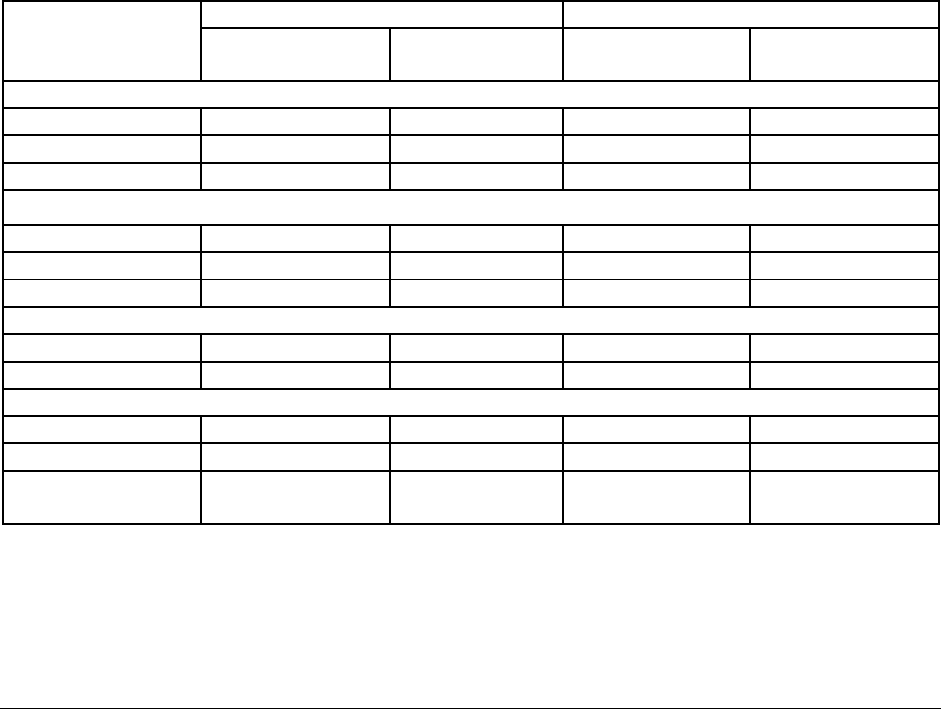
Table 1: Frequency of Adverse Experiences by Body System Reported in ≥1% of Subjects
for Emergency Contraception: Subjects in Controlled Clinical Trials (June 1998)
Body System/
MedDRA
Preferred Term
(WHO/HRP 1998 - Study 92908)
Ho and Kwan, 1993
Levonorgestrel
N = 977 (%)
Yuzpe
N = 979 (%)
Levonorgestrel
N = 410 (%)
Yuzpe
N = 424 (%)
Body, Whole
Abdominal pain
172 (17.6)
205 (20.9)
---
---
Fatigue
165 (16.9)
279 (28.5)
98 (23.9)*
156 (36.8)
Flu syndrome
10 (1.0)
9 (0.9)
---
---
Digestive
Diarrhea
49 (5.0)
64 (6.5)
---
---
Nausea
226 (23.1)*
494 (50.5)
66 (16.1)*
197 (46.5)
Vomiting
55 (5.6)*
184 (18.8)
11 (2.7)*
95 (22.4)
Nervous
Dizziness
109 (11.2)
163 (16.6)
76 (18.5)
98 (23.1)
Headache
164 (16.8)
198 (20.2)
---
---
Urogenital
Breast tenderness
105 (10.7)
118 (12.1)
65 (15.9)
88 (20.8)
Bleeding more
133 (15.6)
116 (11.8)
---
---
Vaginal
hemorrhage
10 (1.0)
12 (1.2)
14 (3.4)
18 (4.2)
*significantly different, P < 0.001

Plan B - Product Monograph Page 9 of 36
Table 2: Frequency of Adverse Experiences by Body System Reported in ≥1% of Subjects
for Emergency Contraception: Subjects in Controlled Clinical Trials (Pooled), March 1999
Body System/MedDRA
Preferred Term
Levonorgestrel
N = 1387 (%)
Yuzpe
N = 1403 (%)
Body, Whole
Fatigue
263 (19.0)*
435 (31.0)
Digestive
Nausea
292 (21.1)*
691 (49.3)
Vomiting
66 (4.8)*
279 (19.9)
Nervous
Dizziness
185 (13.3)*
261 (18.7)
Urogenital
Breast tenderness
170 (12.3)
206 (14.7)
Spotting/bleeding
24 (1.7)
30 (2.1)
*significantly different, P < 0.001
The Plan B
®
OTC Label Comprehension Study
18
was conducted to evaluate whether Plan B
®
can
be used safely and effectively without oversight by a licensed medical practitioner. A total of
540 women used the study product. No serious adverse events were reported. The findings of
the study, compared to those of the Pivotal study, indicate that the pattern of adverse events does
not change when the product is provided in a non-prescription setting. (Table 3)
Table 3: Adverse Events in OTC Actual Use Study and WHO Pivotal Clinical Trial
% Subjects Reporting Adverse Events
Adverse Event
OTC Actual Use Study
WCC/FHI 2002
N = 540
WHO/HRP 1998
Study 92908
N = 979
Abdominal Pain
14.3
17.6
Asthenia (Fatigue)
8.0
16.9
Headache
11.3
5.6
Nausea
12.4
23.1
Vomiting
1.2
5.6
Metrorrhagia
4.3
--
Dizziness
3.7
11.2
All other
29.6
13.5
Plan B
®
single dose administration (Levonorgestrel 1.5 mg tablet)
In a double-blind pivotal trial, the safety profile of levonorgestrel was compared following the
administration of a single dose of 1.5 mg or two doses of 0.75 mg at a 12-hour interval (study

Plan B - Product Monograph Page 10 of 36
97902). A total of 2,756 women used the study product. There was no statistically significant
difference in the incidence of the adverse events between the two levonorgestrel groups.
Table 4: Frequency of Adverse Experiences by Body System Reported in ≥1% of Subjects
for Emergency Contraception: Subjects in Study 97902
Adverse Event
Levonorgestrel
One dose of 1.5 mg
N = 1379 (%)
Levonorgestrel two tablets of 0.75 mg
Administered at a 12-hour interval
N = 1377 (%)
Lower abdominal pain
183 (13.3)
198 (14.4)
Fatigue
184 (13.3)
182 (13.2)
Diarrhea
53 (3.8)
44 (3.2)
Nausea
189 (13.7)
199 (14.5)
Vomiting
19 (1.4)
19 (1.4)
Dizziness
132 (9.6)
126 (9.2)
Headache
142 (10.3)
130 (9.4)
Breast tenderness
113 (8.2)
115 (8.4)
Bleeding more
426 (30.9)
426 (30.9)
Delay of menses
more than 7 days*
61 (4.5)
61 (4.5)
* The denominator for “Delay of menses more than 7 days” is 1359 and 1353, compared to
1379 and 1377 for the other adverse experiences listed in the table.
Less Common Clinical Trial Adverse Drug Reactions (<1%)
Body as a whole: Back muscular pain, influenza, light headedness, numbness in lower
extremities, migraine NOS, redness arms, redness of the chest, tiredness
Cardiovascular: Palpitations
Ear and Nose: Otitis
Gastrointestinal: Acute appendicitis, air in stomach, constipation, polydipsia
Hematology: High platelets counts, low haemoglobin (anaemia hypochromic)
Immunology: Acute tonsillitis, otitis, pharyngitis
Metabolic: Blood glucose increased
Musculoskeletal: Muscle twitching, leg cramps
Psychiatric: Weeping
Plan B - Product Monograph Page 11 of 36
Reproductive: Corpus luteum cyst or haematoma, ectopic pregnancy termination, late menses,
vaginal mycosis
Respiratory: Coughing, rhinitis, sinusitis NOS
Skin and appendages: Acne, acne aggravated, chapped lips, rash
Urinary: Blood in urine, cystitis, ketone positive in urine, urinary protein increased, white
blood cells positive in urine
Post-Market Adverse Drug Reactions
The following adverse reactions have been identified during post-approval use of Plan B
®
.
Because these reactions are reported voluntarily from a population of uncertain size, it is not
always possible to reliably estimate their frequency or establish a causal relationship to drug
exposure.
Gastrointestinal disorders: Abdominal distension, abdominal pain, diarrhoea, nausea, vomiting
General disorders and administration site conditions: Fatigue
Musculoskeletal and connective tissue disorders: Back pain
Nervous system disorders: Dizziness, headache
Pregnancy, puerperium and perinatal conditions: Unintended pregnancy
Reproductive system and breast disorders: Breast tenderness, dysmenorrhoea, menorrhagia,
menstruation delayed, menstruation irregular, oligomenorrhoea, pelvic pain, uterine
haemorrhage, vaginal discharge, vaginal hemorrhage.
DRUG INTERACTIONS
Overview
There are no published drug interaction studies of levonorgestrel. Contraceptive steroids are
known to be sensitive to anticonvulsants, griseofulvin, rifampicin, and certain other antibiotics
(decreased efficacy) and acetaminophen
3
. As the efficacy of Plan B
®
may be decreased in cases
of concomitant intake of these drugs, it is recommended that patients perform a pregnancy test if
menses is delayed beyond 1 week.

Plan B - Product Monograph Page 12 of 36
Drug-Drug Interactions
Anti-HIV Drugs: The metabolism of levonorgestrel is enhanced by the concomitant use of liver
enzyme inducers, mainly CYP3A4 enzyme inducers. Concomitant administration of efavirenz
has been found to reduce plasma levels of levonorgestrel (AUC) by around 50%
24, 25, 26
. A
similar reduction is suspected with the concomitant administration of ritonavir.
Anticonvulsant Drugs: There was a marked decrease in the AUC of levonorgestrel following 12
weeks of treatment with phenytoin and carbamazepine (42% and 40%, respectively)
5
. In
contrast, sodium valproate had no detectable effect. These results are consistent with the known
effects of the drugs on liver enzyme induction. A number of reports exist in the literature on oral
contraceptive failures in women using certain anticonvulsants, most commonly phenytoin,
primidone and phenobarbital.
Antibiotics (see below for rifampicin and rifabutin): No consistent effect has been found in
formal pharmacokinetic studies
3
of a number of antibiotics (including ampicillin, cotrimoxazole,
tetracycline, the quinolone temafloxacin, and the macrolide clarithromycin) on plasma
concentrations of steroids, in particular ethinyl estradiol. It is impossible at the present time to
evaluate fully the potential impact of antibiotics on efficacy based on the literature.
Rifampicin/Rifabutin: Rifampicin is a potent enzyme inducer and, as with anticonvulsant drugs,
there is a molecular basis for expecting an interaction with contraceptive steroid efficacy. Oral
contraceptive failures, menstrual abnormalities, and low progestin levels have been shown in
subjects being treated simultaneously with oral contraceptives and rifampicin
13
. The metabolism
of levonorgestrel is enhanced by the concomitant use of rifabutin.
Acetaminophen: Paracetamol is metabolized primarily by conjugation with sulfuric and
glucuronic acids and hence has the potential for interfering with ethinyl estradiol metabolism.
However, a similar effect on levonorgestrel was not shown in the same study
15
.
Ulipristal acetate: Ulipristal acetate is a progesterone receptor modulator that may interact with
the progestational activity of levonorgestrel. Therefore the concomitant use of levonorgestrel and
drugs containing ulipristal acetate is not recommended.
Drug-Food Interactions
No formal pharmacokinetic studies of the effect of food have been performed. Efficacy is
presumed to be independent of the timing of meals since no instruction on timing of dosing
relative to meals was provided to the participants in the primary clinical trials supporting the
indication.
Drug-Herb Interactions
The concomitant use of Hypericum perforatum (St-John’s wort) can decrease the efficacy of
Plan B
®
.

Plan B - Product Monograph Page 13 of 36
Interactions with other herbal products have not been established.
Drug-Laboratory Interactions
Use of oral contraceptives can modify the results of laboratory tests. Lab tests should therefore
be done prior to dosing or more than 3 days after dosing to avoid misinterpretation of the results.
Pathologists should be advised about oral contraceptive therapy when specimens obtained from
Pap smears are submitted for examination.
DOSAGE AND ADMINISTRATION
Dosing Considerations
Plan B
®
can be administered at any time during the menstrual cycle.
Recommended Dose and Dosage Adjustment
One tablet of Plan B
®
(levonorgestrel 1.5 mg) should be taken orally as soon as possible but
within 72 hours after unprotected intercourse. The total dosage for one complete regimen of Plan
B
®
consists in a single dose of 1.5 mg levonorgestrel.
The patient should be instructed to contact her health care professional if she vomits in the first
two hours after taking the dose of medication. An additional dose may be administered, based
on the judgment of the health care professional. In clinical studies, of the 55 women who
vomited as a result of taking Plan B
®
, 40 took a replacement dose. Statistical analysis showed
that the replacement dose did not increase efficacy significantly. If vomiting occurs as a result of
taking Plan B
®
, it is possible that sufficient quantities of the hormone have been absorbed, as the
maximum blood level after oral consumption is reached in about 1.6 hours. If vomiting occurs,
for other reasons (such as the flu), or if the pills are visible in the emesis, a replacement dose
may be warranted.
The patient should be counselled to abstain of or use an alternative method of contraception
(e.g., diaphragm or condom) until the next menstrual cycle. Most patients will have their next
menstrual period at the expected time or within a week of the expected time. If a normal period
is delayed beyond 1 week, the patient’s pregnancy status should be confirmed with a pregnancy
test and follow-up with a health professional.
OVERDOSAGE
For management of a suspected drug overdose, contact your regional Poison Control Centre.
There are no data on overdosage of Plan B
®
; however, it is anticipated that the incidence and
severity of nausea and vomiting and of menstrual cycle disturbances may be increased. In case

Plan B - Product Monograph Page 14 of 36
of overdose or accidental ingestion by children, treatment is generally not required, but the
patient should be closely observed by the physician and gastric lavage may be employed if
considered necessary.
ACTION AND CLINICAL PHARMACOLOGY
Mechanism of Action
Emergency contraceptives are intended to be used after known or suspected contraceptive failure
or unprotected intercourse. They are not effective if a woman is already pregnant. Plan B
®
(levonorgestrel) is believed to act as an emergency contraceptive principally by preventing
ovulation or by inhibiting fertilization (by altering tubal transport of sperm and/or ova). It may
also prevent implantation (by altering the endometrium). It is not effective once the process of
implantation has begun.
Pharmacokinetics
Absorption and Distribution:
The absolute bioavailability of Plan B
®
in humans has not been specifically investigated.
However, levonorgestrel is reported in the literature to be rapidly and completely absorbed
following oral administration and does not undergo first pass metabolism.
Single dose Studies:
In WCC-PK 001, investigators administered single doses of 0.75 mg tablets to 16 healthy young
women. The same subjects received levonorgestrel 0.75 mg as an oral suspension, prepared
immediately prior to dosing. The rate and extent of absorption following administration of the
suspension were lower than those measured following administration of the tablet (mean C
max
of
7.52 ± 4.14 ng /mL occurring on average 2.8 ± 1.1 hours post-dose for the suspension versus
14.1 ± 7.7 ng /mL and 1.6 ± 0.7 hours post-dose for the tablets). This pattern was observed in
14 of 16 subjects. (The lower bioavailability of the oral suspension is attributed to the larger
particle size in the micronized drug substance as compared with that in the tablet.)
The volume of distribution (Vd) was estimated to be 260.0 L. Serum levels declined with a
mean terminal half-life of 24.4 ± 5.3 hours following administration as a tablet and 27.3 ± 6.3
hours following administration as a suspension.
Results for the three published studies were similar to one another and also to results for the tablet
in the WCC-sponsored study (Table 5). Reported half-lives were shorter in the published single
dose studies, approximately 13 to 14 hours. The duration of sampling was shorter.

Plan B - Product Monograph Page 15 of 36
Table 5: Summary of Pharmacokinetic Parameter Values for Studies
Study
N
Dose
Mean (± S.D.)
C
max
(ng/mL)
T
max
(h)
T
α
(h)
T
β
(h)
Vd
(L)
CL
(L/h)
AUC
0-∞
(ng/mL/h)
WCC-Sponsored Study of Proposed Commercial Formulation
WCC-
PK 001
16
0.75 mg
14.1 ± 7.7
1.6 ± 0.7
–
24.4 ± 5.3
260.0
7.7 ± 2.7
123.1 ± 50.1
Other Studies Performed with Gedeon Richter Formulation
He 1990
10
0.75 mg
11.2 ± 3.4
1.9 ± 0.6
1.3 ± 0.6
13.3 ± 3.7
115 ± 41
6.1 ± 1.9
124 ± 43
Landgren
1989
10
0.75 mg
16.0
–
–
14.5
–
–
–
Shi 1988
6
0.75 mg
9.0 ± 2.2
2 ± 4
–
8.9 ± 1.9
88.6 ±25.6
7.2 ± 2.7
116 ± 41
Multiple Dose Study:
One study
16
, also provided results for seven-day dosing of six healthy young women. Serum
levels on Day 7 were found to be somewhat lower than Day 1 levels (mean C
max
value of 9.0
ng/mL for Day 1; 5.3 ng/mL mean C
max
for Day 7 corrected values), and a longer half-life was
reported (mean 12.6 hours on Day 7). Steady state was obtained on the fourth day, with no
unanticipated accumulation.
Orally administered levonorgestrel is found in breast milk at levels approximating a plasma/milk
ratio of 100:15.
Metabolism: Following a single oral dosage, levonorgestrel does not appear to be extensively
metabolized by the liver. The primary metabolites are 3a,5b- and 3a,5a-tetrahydrolevonorgestrel
with 16b-hydroxynorgestrel also identified. Together, these account for less than 10% of parent
plasma levels. Urinary metabolites hydroxylated at the 2a and 16b positions have also been
identified. Small amounts of the metabolites are present in plasma as sulfate and glucuronide
conjugates.
Special Populations and Conditions
Age Effects: Due to the age range of women participating in these studies (19-44 years), and
given that the target population for levonorgestrel emergency contraception is comparable, age
effects have not been evaluated.
Race: Pharmacokinetic parameters have been summarized separately by ethnicity (Table 6).
Ethnicity was not stated in the three published studies. Two studies were conducted in China,
and presumably, all 16 subjects were Chinese. Similarly, the ten subjects participating in
Sweden are all assumed to be Caucasian. There is a suggestion of lower concentrations in Asian
subjects. These observations should be interpreted with caution, however, as in the U.S.-based
study, there was only one Asian subject, and the assay methodology in the other studies differed.
Plan B - Product Monograph Page 16 of 36
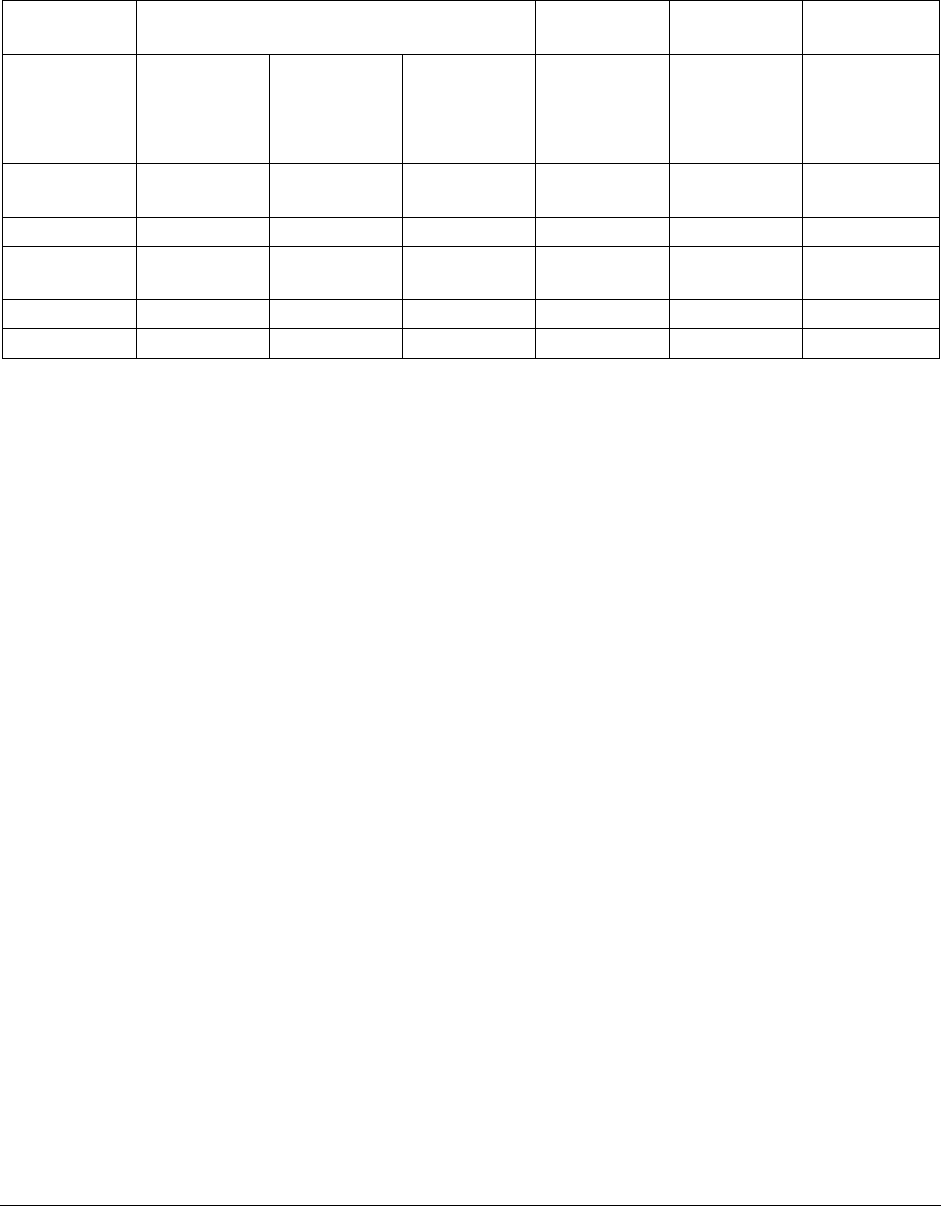
Table 6: Summary of Levonorgestrel 0.75 mg Single Dose Tablet Pharmacokinetic
Parameter Values by Ethnicity
Parameters
WCC-PK 001
Landgren,
1989
He, 1990
Shi, 1988
Caucasian
(U.S.)
(N=9)
Black
(U.S.)
(N=6)
Asian/Pacifi
c Islander
(U.S.)
(N=1)
—
(Sweden)
(N=10)
—
(China)
(N=10)
—
(China)
(N=6)
Cmax
(ng/mL)
15.9
12.2
9.4
16.0
11.2 ± 3.4
9.0 ± 2.2
Tmax (h)
1.8
1.4
1.3
—
1.9 ± 0.6
2-4
AUC0-∞
(ng/mL/h)
131.5
120.7
62.5
—
124 ± 43
116 ± 41
Half life (h)
24.6
24.5
22.9
14.5
13.3 ± 3.7
8.9 ± 1.9
CL (L/hr)
6.4
7.2
12.0
—
6.1 ± 1.9
7.2 ± 2.7
Hepatic and Renal Insufficiency: No formal pharmacokinetic studies have been conducted in
patients with renal or hepatic impairment. Since the product is administered as a single course of
treatment there is no concern about the potential accumulation that might occur with chronic
dosing in patients with hepatic or renal impairment.
STORAGE AND STABILITY
Store Plan B
®
tablet between 15°C and 30°C (59-86 °F). Protect from high humidity.
DOSAGE FORMS, COMPOSITION AND PACKAGING
How Supplied: Almost white, round, flat tablets of about 8 mm diameter with an impressed
mark of « G00» on one side.
Packaging: Plan B
®
tablet (1.5 mg of levonorgestrel) are available in PVC/aluminum foil blister
packages of one tablet each, which is permanently sealed in a double-layer, four-panel outer
package.
Composition: The Plan B
®
tablet contains 1.5 mg of a single active steroid ingredient,
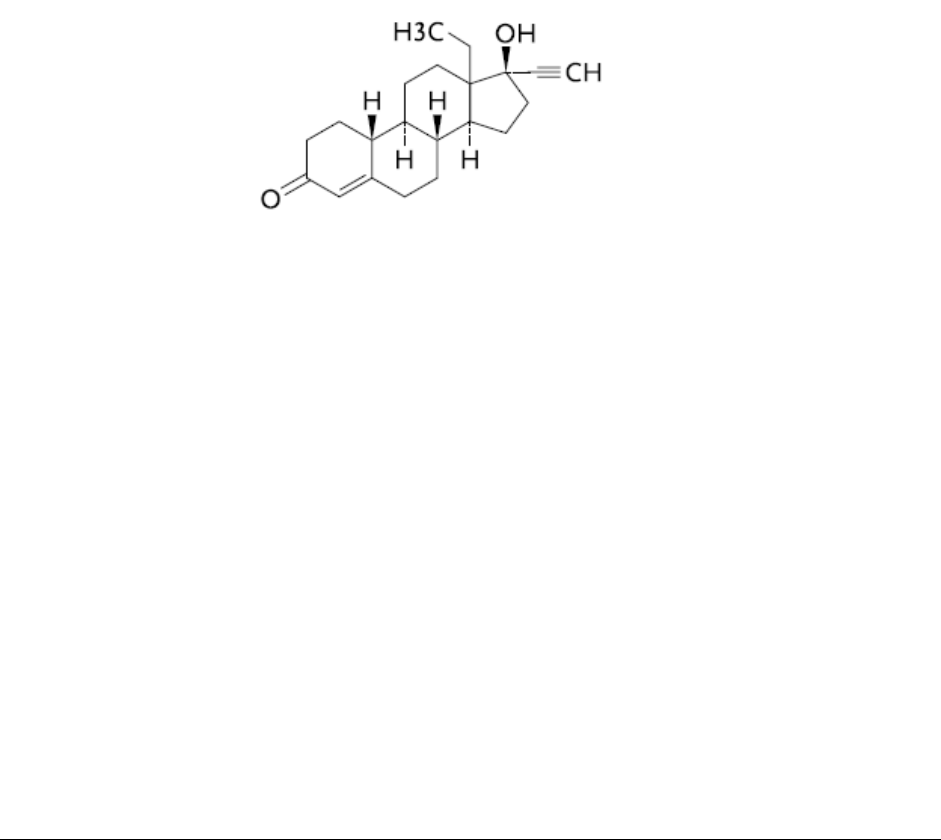
levonorgestrel [18,19-Dinorpregn-4-en-20-yn-3-one,13-ethyl-17-hydroxy–(17α)-(-)-], a totally
synthetic progestogen. The inactive ingredients present are colloidal silicon dioxide, potato
starch, magnesium stearate, talc, cornstarch, and lactose monohydrate.
Plan B - Product Monograph Page 17 of 36
PART II: SCIENTIFIC INFORMATION
PHARMACEUTICAL INFORMATION
Drug Substance
Proper name: Levonorgestrel (USAN), Progestin (INN, BAN)
Chemical name: 18,19-Dinorpregn-4-en-20-yn-3-one, 13-ethyl-17-hydroxy-(17α)-(-)-
Molecular formula: C
21
H
28
O
2
Molecular mass: 312.45
Structural formula:
Physicochemical properties:
Description: White or almost white crystalline powder.
Solubility: Practically insoluble in water, soluble in chloroform, sparingly soluble in
ethanol and methylene chloride.
Melting Point: 232 - 239 °C

Plan B - Product Monograph Page 18 of 36
CLINICAL TRIALS
Plan B
®
divided dose regimen
Study demographics and trial design
Two well-controlled studies have evaluated a two tablet regimen of levonorgestrel 0.75 mg
tablets specifically for emergency contraception. Both were randomized trials comparing the
levonorgestrel regimen to the standard Yuzpe regimen of combined high-dose oral
contraceptives.
Pivotal Study (Study 92908)
The Pivotal Study was conducted between 1995 and 1997 at 21 clinical sites in 14 countries on 5
continents. The earlier controlled clinical trial by Ho and Kwan showed that levonorgestrel alone
was at least as effective as a regimen combining ethinyl estradiol and levonorgestrel (the Yuzpe
regimen), and was associated with significantly lower incidence of nausea, vomiting, and other
side effects. The sample size on the Ho and Kwan study of 834 women was too small, however, to
detect a difference in efficacy of levonorgestrel and hence the pivotal trial was commenced.
In the Pivotal Study, a total of 1,998 women were recruited, including 1,001 in the levonorgestrel
group and 997 in the Yuzpe group; between 42 and 200 women were enrolled at each site. Of the
enrolled women, the final pregnancy status was unknown in 43 women who were lost to follow-
up. The 100 protocol violations included 4 women who were found after discontinuation to have
been pregnant at admission (1 in the levonorgestrel group and 3 in the Yuzpe group) and 5 who
were pregnant at discontinuation but whose pregnancy status at admission was unknown (two in
the levonorgestrel group and 3 in the Yuzpe group). These latter 5 women were excluded from the
primary efficacy analysis population, which thus included 976 women in the levonorgestrel group
and 979 women in the Yuzpe group. Among these 1,995 women, an additional 698 used the
treatment imperfectly.
Ho and Kwan Study
In the Ho and Kwan study, 440 women were recruited into each group. Of these, 46 never
returned after admission and were excluded from the analysis. No women were found to have
been pregnant at admission.
OTC Actual Use Study
An OTC (over-the-counter) Actual Use study
18
was also conducted to evaluate whether Plan B
®
can be used safely and effectively without oversight by a licensed medical practitioner. The study
was conducted in the year 2000 at five affiliates of Planned Parenthood Federation of America
located in geographically diverse regions of the United States and at five pharmacies in the US
state of Washington.

Plan B - Product Monograph Page 19 of 36
Study results
Pivotal and Ho and Kwan Studies
No differences in baseline characteristics were detected between groups in the primary efficacy
analysis population in either study. In both studies, the average age was about 27 years, and many
women had previously been pregnant. The ethnic background of the women in the two studies
differed substantially owing to the locations of the study centres. In both studies, about half of the
participants requested emergency contraception because of failure to use a method during
intercourse, and the rest requested it because of method failure (such as a ruptured condom).
Distribution of the day of intercourse relative to ovulations was similar between groups within
each study. In both studies, the distribution was skewed such that more women had intercourse
before ovulation than after. In each study, the two groups were also similar in the timing of
intercourse and treatment received. In the Pivotal Study, just under half started treatment within
24 hours after intercourse, whereas in the Ho and Kwan study, almost two-thirds did so.
In the Pivotal Study, 31 women (3.2%) became pregnant in the Yuzpe group and 11 (1.1%)
became pregnant in the levonorgestrel group. The relative risk of pregnancy in the Yuzpe group
was 2.8 times higher than that of the Plan B
®
group, with a lower one-sided 95% confidence
bound of 1.53. Since the lower confidence bound was greater than 0.5, the levonorgestrel regimen
was deemed to be as effective as the Yuzpe regimen. The fact that this bound was greater than 1
was considered strong evidence that the levonorgestrel regimen was more effective than the
Yuzpe regimen.
Treatment should not be delayed as efficacy may decline with an increased interval between
intercourse and the start of treatment. Efficacy is greatest when treatment is taken within 24
hours of unprotected intercourse (95% of expected pregnancies prevented), decreasing somewhat
during each subsequent 24 hour period (prevents 61% of expected pregnancies when taken
during the 48 to 72 hour period).
In the Ho and Kwan study, 12 women (2.9%) became pregnant in the levonorgestrel group and 15
(3.5%) became pregnant in the Yuzpe group.
Ectopic Pregnancy
Ectopic pregnancy is a potential risk of progestin-only contraception; however, the reports of
ectopic pregnancies are sparse. They do not appear to occur more in women using emergency
contraception than in the untreated population. The controlled and uncontrolled trials have been
reviewed for possible ectopic pregnancies. If ectopic pregnancies occurred in up to 10% of
pregnancies as predicted by the literature
13
, 3 ectopic pregnancies would have been seen in the
five studies conducted by WHO/HRP. None were observed. Among the 30 pregnancies reported
in the 30 other small studies of levonorgestrel 0.75 mg reviewed, no ectopic pregnancies were
reported. One ectopic pregnancy is noted in the literature. There appears to be no increase in the
rate of ectopic pregnancy after use of levonorgestrel for emergency contraception. There were
also no reports of congenital anomalies in any of the clinical studies.
Plan B - Product Monograph Page 20 of 36
OTC Actual Use Study
In the OTC Actual Use study, 665 women were screened, 585 were enrolled in the study, and 540
are known to have used the product at least once. The primary objective was to estimate the
frequency of contraindicated and incorrect uses of Plan B
®
when dispensed under simulated OTC
conditions. Secondary objectives were to estimate the incidence of repeat use, pregnancy and
adverse events. The label was written to be understood by women of low literacy. Women
received no medical screening or counselling prior to receiving the product.
The reproductive and contraceptive histories were similar across the study populations and
consisted of a wide range of demographic characteristics representative of the female population
of the USA. About 68% of the enrolled population had never been pregnant before their first
screening and more than 80% had no living children. A total of 92% reported having used a
contraceptive method in the month before admission and 60% indicated that they had had sex
without contraception at least once in that month. About 16% reported irregular menstrual
cycles.
Contraindicated use occurred in 1.3% and incorrect use occurred in 7% of users. Eight subjects
used the product twice and two used it three times during the course of the study; the reason for
each repeat use was consistent with the indications for use described on the product label. Ten
of the product users (1.9%) were noted to be pregnant during the study. Adverse events reported
by more than 5% of the subjects were abdominal pain, headache, nausea, and asthenia. These
findings were consistent across subgroups of the study populations defined by age, race,
ethnicity, and education level. No serious adverse events occurred.
Under simulated OTC conditions, the incidence of contraindicated use was extremely low, the
incidence of incorrect use was also very low, and repeat use was very low. The pregnancy rate
and pattern of adverse events were consistent with the findings of previous studies of the
product.
Plan B
®
single dose regimen
Comparative Bioavailability Study
A single dose crossover comparative bioavailability study of Levonorgestrel 1 x 1.5 mg tablet
versus Plan B
®
2 x 0.75 mg tablets was performed in 30 healthy adult female volunteers under
fasted conditions. A summary of the bioavailability data is tabulated below.
Plan B - Product Monograph Page 21 of 36
Table 7: Pharmacokinetic parameters and treatment comparisons
Levonorgestrel
From measured data
Uncorrected for potency
Geometric Mean
Arithmetic Mean (CV %)
Parameter
Test
*
Reference**
% Ratio of
Geometric
Means
Confidence Interval
(90%)
AUC
0-72h
(ng-hr/mL)
228.94
†
268.68 (73.20)
223.01
244.83 (45.15)
102.10%
93.46% - 111.55%
AUC
T
(ng-hr/mL)
253.21
†
294.80 (70.83)
247.35
269.81 (43.46)
101.51%
93.05% - 110.74%
AUC
I
(ng-hr/mL)
264.65
†
307.51 (71.04)
257.26
279.67 (42.66)
102.01%
93.31% - 111.52%
C
max
(ng/mL)
17.35
19.14 (50.48)
17.08
18.14 (33.85)
101.60%
91.97% - 112.25%
T
max
§
(h)
1.67 (1.00 – 4.00)
1.33 (1.00-3.00)
T
½
□
(h)
27.49
†
(20.35)
26.40
(18.26)
*
Levonorgestrel 1.5mg Tablet; administered as 1 x 1.5mg
**
Plan B
®
0.75mg Tablet; administered as 2 x 0.75mg
¤ AUC
T
represents AUC
0-120h
§
Expressed as the median (range) only
□
Expressed as the arithmetic mean (CV%) only
†n=29
Pivotal clinical trial (Study 97902)
Study Demographics and Trial Design
A double blind, randomized, multicentre study evaluated and compared the efficacy and safety
of levonorgestrel for emergency contraception following the administration of one single dose of
1.5 mg, as well as when two doses of one 0.75 mg each were given at a 12-hour interval. This
study was done in 15 family-planning clinics in China, Finland, Georgia, Hungary, India,
Mongolia, Slovenia, Sweden, Switzerland, and the UK between 1998 and 2001. Each centre
recruited between 41 and 149 subjects.

Plan B - Product Monograph Page 22 of 36
A total of 2,756 healthy women, who needed emergency contraception were randomly allocated
into one of the two dose groups, with 1379 to receive a single dose of 1.5 mg levonorgestrel and
1377 to receive two doses of 0.75 mg levonorgestrel. Overall, 41 subjects (1.5%) were lost to
follow-up and the outcome of treatment was unknown. Of these, 22 were in the single dose
levonorgestrel group and 19 were in the two-dose levonorgestrel group. Three women (0.1%)
were found to have had unprotected intercourse after the expected date of menses were
considered withdrawn as they had been erroneously treated (i.e., they had no need for EC). Of
these 3 subjects, 1 was in the two-dose levonorgestrel group and 2 were in the single dose
levonorgestrel group. The intent to treat population was therefore 1356, in both groups.
There were 156 protocol violations among 144 subjects. These were primarily further acts of
unprotected intercourse (31 in the single dose group and 30 in the two-dose group) and use of
rhythm methods in current cycle (23 in the single dose group and 37 in the two-dose group).
Study Results
Baseline characteristics were similar for subjects randomized for each group. The mean age was
27, the mean weight was 56 kg, the mean height was 163 cm, and the mean duration of
menstrual flow was 5 days. The mean cycle length was 29.2 days in the single dose group and
29.3 days in the two-dose group. In both groups 54% were Chinese, 34% were Caucasian and
12% were Asian or Black.
In the single dose group 53.5% of subjects requested emergency contraception because of failure
to use a method during intercourse, and 36.4% requested it because of method failure.
Corresponding figures for the two-dose group were 50.6% and 39.5%, respectively.
In the single dose group 45.9% of subjects requested treatment within 24hr, 27.8% within 48 hr,
14.7% within 72 hr, 6.4% within 96 hr and 4.6% requested treatment after 97 hrs.
Corresponding figures for the two-dose group were 42.2%, 26.6%, 18.4%, 7.4% and 4.6%,
respectively.
The fraction of prevented pregnancies was 81.9% (CI: 72.1% to 88.9%) in the levonorgestrel
single group and 77.3% (CI: 66.3% to 85.5%) in the 0.75 mg two-dose group, with a relative risk
of pregnancy of 0.83 (CI: 0.46 to 1.50) for the levonorgestrel single dose group over the 0.75 mg
two-dose group.
The two levonorgestrel regimens studied are highly effective for emergency contraception. Both
studies showed that the 1.5 mg levonorgestrel single dose regimen had a similar effectiveness
with the two doses of 0.75 mg levonorgestrel. There was no significant difference between the
two levonorgestrel groups (p>0.6).
When comparing the efficacy of treatment in women starting the treatment within 3 days of
unprotected intercourse and those starting treatment within a delay of 4 or 5 days, there was a

Plan B - Product Monograph Page 23 of 36
decreasing trend of efficacy found, although the difference in efficacy was not statistically
significant.
For all treatment groups combined, there was a significant trend in pregnancy rates in the 5
successive days from the time of unprotected intercourse in each efficacy set, showing an
increase in pregnancy rates by days elapsed from unprotected intercourse. A similar trend was
found when the two levonorgestrel regimens were combined.
Post-hoc analysis of efficacy by weight:
In clinical trials of products similar to Plan B
®
, a post-hoc analysis
20
of data from two published
papers
21, 22
on the efficacy of levonorgestel 1.5 mg (single dose) and 0.75 mg (two doses 12
hours apart) raised questions concerning the efficacy of single ingredient levonorgestrel
emergency contraceptives in women with higher body weights.
There is limited and inconclusive data on the effect of high body weight/high BMI in the
contraceptive efficacy. In three WHO studies
(1, 19, 23)
no trend for a reduced efficacy with
increasing body weight/BMI was observed (Table 8), whereas in the two other studies
(21, 22)
a
reduced contraceptive efficacy was observed with increasing body weight or BMI (Table 9).
Both meta-analyses excluded intake later than 72 hours after unprotected intercourse and women
who had further acts of unprotected intercourse.
Table 8: Meta-analyses on three WHO studies
1, 19, 23
BMI (kg/m
2
)
Underweight
0 – 18.5
Normal
18.5 - 25
Overweight
25 – 30
Obese
≥ 30
N total
600
3952
1051
256
N pregnancies
11
39
6
3
Pregnancy rate
1.83%
0.99%
0.57%
1.17%
Confidence Interval
0.92 – 3.26
0.70 – 1.35
0.21 – 1.24
0.24 – 3.39
Table 9: Meta-analyses on two studies
21, 22
BMI (kg/m
2
)
Underweight
0 – 18.5
Normal
18.5 - 25
Overweight
25 – 30
Obese
≥ 30
N total
64
933
339
212
N pregnancies
1
9
8
11
Pregnancy rate
1.56%
0.96%
2.36%
5.19%
Confidence Interval
0.04 – 8.40
0.44 – 1.82
1.02 – 4.60
2.62 – 9.09

Plan B - Product Monograph Page 24 of 36
DETAILED PHARMACOLOGY
Pharmacodynamics
Animal in vivo studies
Levonorgestrel is active in standard tests for progesterone activity, acts like a true progestin when
tested for pregnancy maintenance, and is neither estrogenic by itself nor metabolically converted
to an estrogen. Non-clinical studies conducted on the use of levonorgestrel or norgestrel have
shown that levonorgestrel administered subcutaneously or orally mimics the primary properties of
progesterone in that it binds to the progesterone receptor and produces glandular transformation of
the endometrium. In addition, levonorgestrel maintains pregnancy in the appropriate animal
model and delays implantation of the blastocysts. Levonorgestrel also inhibits ovulation,
gonadotropin secretion, and shows contraceptive activity as demonstrated in a number of
biological systems. The qualitative and quantitative parameters for the reproductive
pharmacology of levonorgestrel have been established through extensive studies using rabbits,
rats, and mice.
Levonorgestrel can increase uterine weight but does not bind to the estrogenic receptor and does
not lead to keratinization of the vaginal epithelium. Anti-estrogenic activity against the vaginal
and uterine effects of estrone and estradiol have been demonstrated following parenteral
administration of levonorgestrel. This estrogenic antagonism suggests levonorgestrel acts like
progesterone. However, there is no evidence to support antagonism of estrogen-induced
mammary tumours, uterine overstimulation, or cataracts. Levonorgestrel can bind to the
androgenic receptor and has anabolic androgenic effects such as protecting bone mass from
osteopenia in an animal model. However, levonorgestrel inhibits ovulation at much lower
dosages than were needed to produce androgenic and anabolic effects on suitable target tissues.
In a number of other standard evaluations of endocrine activity, levonorgestrel was devoid of
detectable glucocorticoid, mineralocorticoid, and anti-mineralocorticoid activity in vivo, even
though it binds to the glucocorticoid (aldosterone) receptor.
The qualitative profile of levonorgestrel with respect to sexual function is summarized in Table
10.
Plan B - Product Monograph Page 25 of 36
Table 10: Qualitative Non-clinical Pharmacological Profile of Levonorgestrel with Respect
to Sexual Functions
Test
Activity
Progestin-like activity
In vivo
Active
Receptor binding
Active
Contraception
In vivo
Active
Inhibition of fertility
Inhibition of the gonadotropins
Active
Inhibition of ovulation
Active
Change in the estrous cycle
Active
Androgen-like/anabolic activity
In vivo
Some active/some inactive
Receptor binding
Some active/some inactive
Antiandrogenic activity
In vivo
Inactive
Estrogenic activity
In vivo
Inactive
Receptor binding
Inactive
Estrogen antagonism
In vivo
Active
Receptor binding
Inactive
Pharmacokinetics
The non-clinical pharmacokinetics of levonorgestrel have been investigated pertaining to its
development as a low-dose oral contraceptive. These data indicate low oral bioavailability of
levonorgestrel in animals as compared to man, urinary and biliary excretion of an absorbed dose,
and the formation of conjugated metabolites. The pharmacokinetics of a single elevated dose
have not been investigated in animals.
TOXICOLOGY
The non-clinical toxicology of levonorgestrel or norgestrel has been investigated in a number of
studies in rodents, dogs, and monkeys, including a 7-year repeat administration study in monkeys
and numerous reproductive toxicity studies and mutagenicity studies. All of these studies were
conducted as part of the non-clinical development of norgestrel as a traditional oral contraceptive
or contraceptive implant. This large body of evidence was used to support the approval of oral
contraceptives containing up to 500 μg of norgestrel in combination with ethinyl estradiol or 75
μg alone, where the intended use required chronic administration.
The Toxicology Review Panel of the Special Programme (WHO) has reviewed the available
animal data on levonorgestrel and has concluded that the toxicological and teratological data
Plan B - Product Monograph Page 26 of 36
from animal studies on levonorgestrel provide a sufficient indication that levonorgestrel may be
considered safe for use as an implantable contraceptive. Toxicity studies are summarized in
Table 11.
Table 11: Previously Reviewed FDA Toxicity Studies
Title
Date
Dose of Wy-3707
Duration
Studies in Rodents
Toxicity Study of Norgestrel in Combination
with Ethinyl Estradiol in Mice (10: and 20:1
ratios)
0.0002 and 0.0004% of diet
81-83 weeks
Toxicity Studies in Rodents
6/12/62
0.0125, 0.025 mg/kg
7.5 weeks
Sub-acute Toxicity of Wy-3707 Alone,
Mixtures of Wy-3707 with Ethinyl Estradiol,
Ethinyl Estradiol Alone in Rats
6/12/64
0.01, 0.1,0.2 mg/kg
7-10 weeks
Toxicity Study of Wy-3707 in Combination
with Ethinyl Estradiol in Rats (Radios of
2:1and 5:1)
7/1/66
0.0001 - 0.0025 mg/kg
36-38.5 weeks
Toxicity Study of Wy-3707 in Combination
with Ethinyl Estradiol in Rats (Radios of 25:1,
20:1, and 2:1)
7/1/66
0.00025, 0.005, 0.001 mg/kg
56 weeks
Chronic Toxicity Study of Wy-3707 in Rats
7/1/66
0.001, 0.01, 0.05, 0.1 mg/kg
62.5-80 weeks
Toxicity Studies of Wy-3707 and Ethinyl
Estradiol in Rats
7/1/66
0.0001 - 0.001 mg/kg
99.5-100 weeks
Toxicity Studies of Wy-3707 in Combination
with Ethinyl Estradiol in Rats (Ratio of 10:1)
7/1/66
0.000 - 0.01 mg/kg
99-100 weeks
Studies in Dogs
Sub-acute Toxicity in Dogs
6/2/64
1, 10, 50 mg/kg
6 weeks
Sub-acute Toxicity Studies of Wy-3707 Alone
and Mixtures of Wy-3707 and Ethinyl Estradiol
Alone in Dogs
6/17/64
1, 10, 50 mg/kg
6 weeks
Chronic Toxicity Study of Wy-3707 in Dogs
10/14/65
1, 10, 20 mg/kg
52-52.5 weeks
Toxicity Studies of Wy-3707 in Combination
with Ethinyl Estradiol in Dogs (Ratio of 10:1)
7/1/66
1 or 2 mg/kg
54 and 104
weeks
Toxicity Studies of Wy-3703 and Wy-3707 in
Combination With Ethinyl Estradiol in Dogs
(Ratio of 20:1)
7/1/66
2 or 4 mg/kg
25-36 weeks
Toxicity Studies of Wy-3707 and Ethinyl
Estradiol in Dogs
7/1/66
0.01, 0.1, 0.2, 1.0 mg/kg
54, 64 and
100/104 weeks
Toxicity Studies of Norgestrel in Dogs
~1977
0.1, 0.25 mg/kg
7 years
Study in Primates
Toxicity Study of Norgestrel in Rhesus
Monkeys
~1977
0.02, 0.1, 0.5 mg/kg
7 years

Plan B - Product Monograph Page 27 of 36
Acute Toxicity Studies: In single dose studies, the LD
50
for norgestrel was > 5000 mg/kg dosed
orally in rodents and dogs. Multiple dose studies in mice for 18 months had no effect. In rats,
other than exaggerated physiological effects expected with high doses of estrogens, there was
little evidence of drug toxicity. Findings included decreased pituitary weights, secretory changes
in the cervix and vagina, and endometrial hyperplastic changes. Growth rate and food
consumption were depressed among norgestrel-treated animals at high doses.
Chronic Toxicity Studies: Data from the first seven years of a ten-year repeat administration
study in Rhesus monkeys with d,l-norgestrel was collected to support clinical use as an oral
contraceptive. Sixty-four female Rhesus monkeys (16/group) were administered norgestrel 0
mg, 0.02 mg, 0.1 mg, and 0.5 mg/kg/day as a dietary admixture for 21 consecutive days followed
by seven days of a non-dosing period (28-day cycle). There were no norgestrel-related effects
on mortality, behaviour, mean body weights, endocrine function, or ophthalmoscopic exams. A
number of minor dose related changes were noted in hematocrit, Stypven Times, activated partial
thromboplastin times, fibrinogen concentrations, and urinary output of 17-
hydroxycorticosteroids. There were no findings in gross or histopathologic evaluations
indicative of toxicity, and microscopic examination of cervical smears did not reveal any
evidence of neoplasia. No mammary nodules were palpable at the end of the first, third, sixth,
and seventh years, none of the monkeys had palpable mammary nodules at the end of the sixth
and seventh years, and there was no effect on the number of palpable mammary nodules or the
month when the first mammary nodule was found over the course of seven years. The
conclusion of the U.S. FDA review of this study indicated that administration of norgestrel to
female monkeys for ten years caused essentially no untoward effects.
Carcinogenicity Studies: In CF-LP (MTV
+
) mice administered doses described as low (2-5
times clinical), mid (50-150 times clinical), and high (200-400 times clinical) for 80 weeks, there
was no effect on incidence of tumours in any tissues. In castrated C3HxRIII (MTV
+
) mice
administered 0.5 mg/kg d-norgestrel or 1 mg/kg dl-norgestrel in the diet, the incidence of
mammary tumours was slightly raised in the absence of any effect on latency. In the rat,
administration of norgestrel in the diet (doses not described) for 104 weeks had no effect on the
incidence of tumours.
Genotoxicity: Levonorgestrel revealed no potential for genotoxicity in a standard Ames
Salmonella/Microsome Mutagenicity Test.
Reproductive Toxicity: A large number of reproductive toxicity studies were performed by
repeat dose administration to evaluate the effects on mating fertility, fecundity, post-treatment
recovery of fertility, effects on the estrous cycle, claudogenic effects as well as classic Segment
I, II, and III reproductive studies. Of greatest possible relevance to emergency contraception are
the studies of recovery of fertility and birth defects. In a study in mice treated with up to 50x the
human contraceptive dose, no irreversible impairment of fertility was noted.
Segment II studies were given by repeat dose during organogenesis. At the levels required to
maintain pregnancy, virilizing effects were noted and were considerably greater than those of
progesterone. Two of 439 fetuses from dams treated at these levels of norgestrel were deformed;

Plan B - Product Monograph Page 28 of 36
one fetus from a dam treated with norgestrel 3 mg subcutaneously had incomplete spinal closure
and one fetus from a dam treated with norgestrel 3 mg orally had a poorly developed cranium.
The 88 control fetuses were normal. Occasional deformities appeared in the other progestogen
groups, and were more frequent from spayed mothers. In a study where norgestrel was given
subcutaneously from Days 16 to 19 of gestation, potency in producing virilization in female
fetuses was found to be nearly equal to testosterone propionate and three times greater than
norethindrone acetate. Histological examination showed that norgestrel 0.1 mg/day
subcutaneously was effective, while 10 mg/day orally was ineffective. For a macroscopically
detectable increase in ano-genital distance, 3 mg/day subcutaneously was required.

Plan B - Product Monograph Page 29 of 36
REFERENCES
1. Task Force on Postovulatory Methods of Fertility Regulation. Randomized controlled trial
of levonorgestrel versus the Yuzpe regimen of combined oral contraceptive for emergency
contraception. The Lancet 1998; 352: 428-33.
2. Bracken M.B. Oral contraception and congenital malformations in offspring: a review and
meta-analysis of the prospective studies. Obstet Gynecol 1990 Sep; 76:552-57.
3. Back DJ, Orme ML’E. Pharmacokinetic drug interactions with oral contraceptive. In:
Steroid Contraceptives and Women’s Response. Snow R and Hall P (Ed.). Plenum Press,
New York. 1994; pp 103-12.
4. Brenner PF, Mishell DR, Stanczyk FZ, Goebelsmann U. Serum levels of d-Norgestrel,
luteinizing hormone, follicle-stimulating hormone, estradiol, and progesterone in women
during and following ingestion of combination oral contraceptives containing dl-
Norgestrel. Am J Obstet Gynecol. 1977; 129:133-40.
5. Crawford et al., as summarized by Back and Orme (referenced above), 1994.
6. Family Health International. Plan B Over-the-Counter Label Comprehension Study.
October 4, 2001. Unpublished manuscript. Prepared under contract with Women’s
Capital Corporation for regulatory submissions.
7. Fotherby K. Levonorgestrel: Clinical pharmacokinetics. Clin Pharmacokinet. 1995;
28:203-15.
8. Fotherby K. Pharmacokinetics of gestagens: Some problems. Am J Obstet Gynecol.
1990; 163:323-328.
9. He CH, Shi YE, Liao DL, Zhu YH, Xu JQ, Matlin SA, Vince PM, Fotherby K, Van Look
PF. Comparative cross-over pharmacokinetic study on two types of postcoital
contraceptive tablets containing levonorgestrel. Contraception. 1990; 41:557-67.
10. Ho PC and Kwan MSW. A prospective randomized comparison of levonorgestrel with the
Yuzpe regimen in post-coital contraception. Human Reproduction. 1993; 8:389-392.
11. Hümpel M, Wendt H, Pommerenke G, Weiss C, Speck U. Investigations of
pharmacokinetics of levonorgestrel to specific consideration of a possible first-pass effect
in women. Contraception. 1978; 17: 207-20.
12. McCann MF. Levonorgestrel literature review. July 2, 1998. Unpublished manuscript.
Prepared under contract with Women’s Capital Corporation for regulatory submissions.

Plan B - Product Monograph Page 30 of 36
13. McCann MF. and Potter LS. Progestin-only oral contraception: A comprehensive review.
Contraception. 1994; 50:S1-S198.
14. Nilsson S, Nygren K, Johansson EDB. D-Norgestrel concentrations in maternal plasma,
milk, and child plasma during administration of oral contraceptives to nursing women.
Am J Obstet Gynecol. 1977; 129:178-84.
15. Rogers SM, Back DJ, Stevenson PJ, Grimmer SF, Fotherby K. Paracetamol interaction
with oral contraceptive steroids: Increased plasma concentrations of ethinyloestradiol. Br
J Clin Pharmac. 1987; 23:721-25.
16. Shi YE, Zheng SH, Zhu YH, He CH, Yu PP, Fotherby K. Pharmacokinetic study of
levonorgestrel used as a postcoital agent. Contraception 1988; 37:359-69.
17. Weiner E, Victor A, Johansson E. Plasma levels of d-Norgestrel after oral administration.
Contraception. 1976; 14:563-70.
18. Family Health International. Plan B Over-the-Counter Actual Use Study. September 30,
2002. Unpublished study report. Prepared under contract with Women’s Capital
Corporation for regulatory submissions.
19. WHO, von Hertzen H, Piaggioa G, et al. Low Dose Mifepristone and Two Regimens of
Levonorgestrel for Emergency Contraception: a WHO Multicenter Randomized Trial.
The Lancet 2002; 360:1803.
20. Glasier A, Cameron ST, Blithe D, Scherrer B, Mathe H, Levy D, Gainer E, Ulmann A .
Can we identify women at risk of pregnancy despite using emergency contraception? Data
from randomized trials of ulipristal acetate and levonorgestrel. Contraception. 2011;
84:363-367
21. Creinin MD, Schlaff W, Archer DF, et al. Progesterone receptor modulator for emergency
contraception. A randomized controlled trial. Obstetrics & Gynecology 2006;
108(5):1089-1097
22. Glasier AF, Cameron ST, Fine PM, Logan SJ, et al. Ulipristal acetate versus
levonorgestrel for emergency contraception: a randomised non-inferiority trial and
metaanalysis. Lancet 2010; 375:555-62 (see also Clinical Trials NCT00271583 and
NCT00551616 at http://clinicaltrials.gov/ct2/home)
23. Dada O.A, Emily M. Godfrey EM, Piaggio G, von Hertzen H. A randomized, double-
blind, noninferiority study to compare two regimens of levonorgestrel for emergency
contraception in Nigeria. Contraception 82 (2010) 373–378.

Plan B - Product Monograph Page 31 of 36
24. Carten ML, Kiser JJ, Kwara A, Mawhinney S, Cu-Uvin S. Pharmacokinetic interactions
between the hormonal emergency contraception, levonorgestrel (Plan B), and efavirenz.
Infect Dis Obstet Gynecol 2011; (2012): 1-4.
25. Scarsi KK, Darin KM, Nakalema S, Back DJ, Byakika-Kibwika P, Else LJ, et al.
Unintended pregnancies observed with combined use of levonorgestrel contraceptive
implant and efavirenz-based antiretroviral therapy: a three arm pharmacokinetic evaluation
over 48 weeks. Clin Infect Dis 2016; 62 (6):675-82.
26. Scarsi K, Lamorde M, Darin K, Dilly Penchala S, Else L, Nakalema S, et al. Efavirenz-but
not nevirapine-based antiretroviral therapy decreases exposure to the levonorgestrel
released from a subdermal contraceptive implant [abstract]. J Int AIDS Soc 2014; 17 (4
Supp 3):19484.
IMPORTANT: PLEASE READ
Plan B - Product Monograph Page 32 of 36
PART III: CONSUMER INFORMATION
PLAN B
®
Levonorgestrel Tablet 1.5 mg
This leaflet is part III of a three-part "Product
Monograph" published when Plan B
®
was approved
for sale in Canada and is designed specifically for
Consumers. This leaflet is a summary and will not tell
you everything about Plan B
®
. Contact your doctor
or pharmacist if you have any questions about the
drug.
ABOUT THIS MEDICATION
What the medication is used for:
Plan B
®
is an emergency or backup contraceptive.
Plan B
®
can prevent pregnancy after a contraceptive
accident (such as a broken condom) or when no form of
birth control was used. Treatment is most effective if
used in the first 72 hours (3 days) following unprotected
sex.
Plan B
®
cannot terminate an existing pregnancy.
Although there is no scientific evidence that Plan B
®
would harm a developing embryo, women who are
already pregnant should not use this product.
Plan B
®
should not be used in place of regular
contraception. It does not work as well as most other
contraceptives used correctly.
What it does:
Plan B
®
acts as an emergency contraceptive by
preventing the release of an egg from the ovary, or
preventing sperm and egg from uniting. In addition, Plan
B
®
may prevent the fertilized egg from attaching to the
wall of the uterus. Plan B
®
is not effective once a
pregnancy has started, that is once the fertilized egg has
attached to the wall of the uterus. Plan B
®
does not cause
an abortion.
Plan B
®
can be used following any unprotected act of
sexual intercourse, including:
When no contraceptive has been used
When a contraceptive method may have
failed, including:
o Condom rupture, slippage, or misuse
o Diaphragm or cap dislodgment,
breakage or early removal
o Failure to withdraw before ejaculation
o Miscalculation of the fertile period by
women practising periodic abstinence
o IUD expulsion
o Missed oral contraceptives
o A delay in starting a new packet of
oral contraceptives
o A delay in getting a scheduled
contraceptive injection
In cases of sexual assault (rape)
When it should not be used:
Do not use Plan B
®
if:
you have a confirmed or suspected pregnancy
you are allergic to Levonorgestrel or to any
ingredients in the formulation (see non-medicinal
ingredients below)
you have abnormal vaginal bleeding
What the medicinal ingredient is:
Levonorgestrel
What the non-medicinal ingredients are:
Colloidal silicon dioxide, potato starch, magnesium
stearate, talc, cornstarch and lactose monohydrate.
What dosage forms it comes in:
The package contains one tablet, containing 1.5 mg of
levonorgestrel. The tablet is white, round and marked
G00.
WARNINGS AND PRECAUTIONS
Serious Warnings and Precautions
Plan B
®
provides no protection against
HIV/AIDS and other sexually transmitted
diseases (STDs), such as syphilis, gonorrhoea,
chlamydia, and herpes. If you are worried about
whether you may have been infected with
HIV/AIDS, or other sexually transmitted diseases,
talk to your health professional about your
concerns and ask how you can protect yourself in
the future.
If your period is delayed more than 1 week,
you should contact your doctor and have a
pregnancy test done.

IMPORTANT: PLEASE READ
Plan B - Product Monograph Page 33 of 36
Plan B
®
is for Emergency Contraceptive Use Only
and should not be used on a regular basis. Plan B is
not as effective in preventing pregnancy as most other
birth control methods (e.g. oral contraceptive pills,
IUDs, implants or condoms, etc.). It should not be
relied on for routine birth control by sexually active
women.
Plan B
®
provides only short-term protection against
pregnancy. Sexual activity that takes place later can
still result in pregnancy if no contraceptive is used.
You must abstain from sex or use another barrier
method of birth control until your next normal period
to make sure you do not get pregnant.
In all women, emergency contraception should be
taken as soon as possible after unprotected intercourse.
There is some data that levonorgestrel may be less
effective with increasing body weight or body mass
index (BMI), but these data were limited and
inconclusive. If you have any questions regarding this,
please consult with a health care professional.
BEFORE you use Plan B
®
, talk to your doctor or
pharmacist if you have now or have previously had any
of the following conditions:
Unusual vaginal bleeding that has not yet been
diagnosed
Known or suspected breast cancer
Active liver disease or tumour
You have diabetes
You have hypertension (a heart condition)
You are pregnant or breast feeding
INTERACTIONS WITH THIS MEDICATION
Before you use Plan B
®
, talk to your doctor or
pharmacist if you are taking any of the following
drugs:
Drugs that may decrease the efficacy of Plan B
®
include:
anti-HIV drugs (efavirenz, ritonavir)
anticonvulsant drugs (phenytoin,
carbamazepine, primidone, phenobarbital)
antibiotics (ampicillin, cotrimoxazole,
tetracycline, temafloxacin, clarithromycin)
rifampicin
rifabutin
griseofulvin
Hypericum perforatum (St. John’s wort)
ulipristal acetate
If your period is delayed more than 1 week and you
were taking any of these medications, you should
contact your doctor or pharmacist and have a
pregnancy test done.
Tell your doctor if you have taken Plan B
®
within 3
days of a Pap test, as it may affect your results.
PROPER USE OF THIS MEDICATION
Usual dose:
Plan B
®
can prevent pregnancy if the tablet is taken
within 72 hours (3 days) and preferably within 12
hours after a contraceptive accident or unprotected
sex (sex without birth control). Do not delay starting
treatment. Plan B
®
is more effective the sooner you start
after unprotected intercourse.
Plan B
®
can be taken with a glass of water.
If you vomit within 2 hours of taking Plan B
®
, call your
doctor or pharmacist as you might need another dose.
Important: If more than 72 hours (3 days) have passed
since unprotected sex occurred, Plan B
®
may not be
effective. See your health professional as soon as
possible to discuss other options.
Although the risk of pregnancy is highest in the middle
of the menstrual cycle (possibly as early as day 10 after
the beginning of your last period), pregnancy can occur
at other times during the menstrual cycle. Plan B
®
can
be administered anytime during your monthly cycle if
you are worried about an unwanted pregnancy.
The treatment does not bring on menstruation. You
may experience spotting a few days after taking Plan
B
®
, but this is not a period. Your next menstrual period
should come on time (or a few days early or late). If
your period is delayed more than a week or if you have
any other cause for concern, talk to a health
professional. More than occasional use (more than once
in a menstrual cycle or more than once a month) may
upset your menstrual cycle (period).
If you are sexually active and do not wish to become
pregnant, you should use a reliable method of
contraception on a regular basis. If you want more
information about regular contraceptives or if you are
having trouble using a method, ask your health
professional for help in choosing a method that works
for you.

IMPORTANT: PLEASE READ
Plan B - Product Monograph Page 34 of 36
Overdose:
In case of drug overdose, contact a health care
professional, hospital emergency department or
regional Poison Control Centre immediately, even if
there are no symptoms.
Symptoms of overdose may include nausea, vomiting,
vaginal bleeding, and may lead to menstrual cycle
disturbances.
SIDE EFFECTS AND WHAT TO DO ABOUT
THEM
Plan B
®
can cause temporary side effects in some
women. These side effects generally do not last more
than 24 hours.
Common side effects include:
Nausea: This occurs in about 14 to 23% of
women taking Plan B
®
Abdominal pain: This occurs in about 18%
of women taking Plan B
®
Fatigue: This occurs in about 17% of women
taking Plan B
®
Headache: This occurs in about 17% of
women taking Plan B
®
Dizziness: This occurs in about 11% of
women taking Plan B
®
Breast tenderness: This occurs in about 11%
of women taking Plan B
®
Vomiting: This occurs in about 6% of women
taking Plan B
®
Diarrhea: This occurs in about 5% of women
taking Plan B
®
Irregular menstrual bleeding: Some women
may experience spotting after taking Plan B
®
.
The majority of women will have their next
menstrual period at the expected time or early.
When Plan B
®
is used repeatedly (more than
once in a menstrual cycle, or more than
occasional once a month use), menstrual
changes may occur, including a shorter or
longer cycle and a heavier or lighter period
than normal.
Less common side effects: Migraine or severe
headache, lower abdominal pain, painful menstruation
and vaginal discharge. If the symptoms continue for
more than 48 hours or are severe, see your health
professional.
Delayed menstrual period: If your period is delayed
more than 1 week, you should contact your doctor or
pharmacist and have a pregnancy test done.
SERIOUS SIDE EFFECTS, HOW OFTEN THEY
HAPPEN AND WHAT TO DO ABOUT THEM
Call your doctor immediately if the following
symptoms and signs of serious adverse effects occur:
Itching and rash
Cramping or severe pain in your stomach or belly
prior to your next normal period, since this can be
a warning sign of tubal (ectopic) pregnancy – a
serious medical problem.
Uterine hemorrhage
Vaginal hemorrhage
HOW TO STORE IT
Store Plan B
®
tablet between 15°C and 30°C (59-86 °F).
Protect from high humidity (such as damp storage
conditions).
Keep out of reach of children.

IMPORTANT: PLEASE READ
Plan B - Product Monograph Page 35 of 36
Reporting Side Effects
You can help improve the safe use of health products for
Canadian by reporting serious and unexpected side
effects to Health Canada. Your report may help to
identify new side effects and change the product safety
information.
3 ways to report:
Online at MedEffect:
https://www.canada.ca/en/health-
canada/services/drugs-health-products/medeffect-
canada/adverse-reaction-reporting.html;
By calling 1-866-234-2345 (toll-free);
By completing a Consumer Side Effect
Reporting Form and sending it by:
o Fax to 1-866-678-6789 (toll-free), or
o Mail to: Canada Vigilance Program
Health Canada,
Postal Locator 0701E
Ottawa, ON
K1A 0K9
Postage paid labels and the Consumer Side Effect
Reporting Form are available at MedEffect
(https://www.canada.ca/en/health-canada/services/drugs-
health-products/medeffect-canada/adverse-reaction-
reporting.html).
Note: Contact your health professional if you need
information about how to manage your side effects. The
Canada Vigilance Program does not provide medical
advice.

IMPORTANT: PLEASE READ
Plan B - Product Monograph Page 36 of 36
MORE INFORMATION
This document plus the full product monograph,
prepared for health professionals can be found by
contacting the distributor, Paladin Labs Inc., at:
1-888-867-7426 or at www.paladinlabs.com.
This leaflet was prepared by:
Paladin Labs Inc.
Saint-Laurent, Canada H4M 2P2
Last revised: April 26, 2018.
Owned by:
Foundation Consumer Healthcare, LLC
1190 Omega Drive, Pittsburgh, PA 15205, USA
